March 2017 – Diagnosing pediatric Burkitt Lymphoma in sub-Saharan Africa is difficult, as many tools are currently not available. This study found that measuring Epstein-Barr virus DNA could help with setting the diagnosis, prognosis, and predicting the response.
Plasma Epstein-Barr virus DNA for pediatric Burkitt lymphoma diagnosis, prognosis, and response assessment in Malawi
Kate Westmoreland, Nathan Montgomery, Christopher Stanley, Nader El-Mallawany, Peter Wasswa, Toon van der Gronde, Idah Mtete, Mercy Butia, Salama Itimu, Mary Chasela, Mary Mtunda, Coxcilly Kampani, George Liomba, Tamiwe Tomoka, Bal Dhungel, Marcia Sanders, Robert Krysiak, Peter Kazembe, Dirk Dittmer, Yuri Fedoriw, Satish Gopal
International Journal of Cancer
Find the paper in PubMed or in the International Journal of Cancer
Abstract
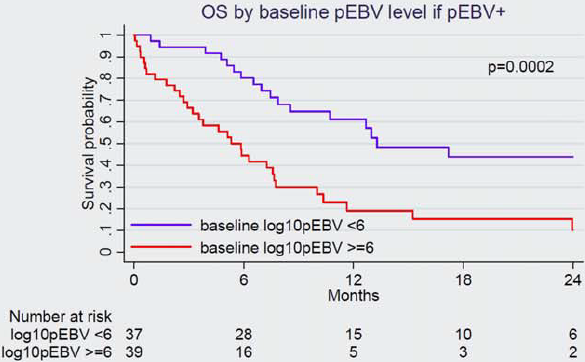
Point-of-care tools are needed in sub-Saharan Africa (SSA) to improve pediatric Burkitt lymphoma (BL) diagnosis and treatment. We evaluated plasma Epstein-Barr virus (pEBV) DNA as a pediatric BL biomarker in Malawi. Prospectively enrolled children with BL were compared to classical Hodgkin lymphoma (cHL) and nonlymphoma diagnoses. Pediatric BL patients received standardized chemotherapy and supportive care. pEBV DNA was measured at baseline, mid-treatment, and treatment completion. Of 121 assessed children, pEBV DNA was detected in 76/88 (86%) with BL, 16/17 (94%) with cHL, and 2/16 (12%) with nonlymphoma, with proportions higher in BL versus nonlymphoma (p < 0.001) and similar in BL versus cHL (p = 0.69). If detected, median pEBV DNA was 6.1 log10 copies/mL for BL, 4.8 log10 copies/mL for cHL, and 3.4 log10 copies/mL for nonlymphoma, with higher levels in BL versus cHL (p = 0.029), and a trend toward higher levels in BL versus nonlymphoma (p = 0.062). pEBV DNA declined during treatment in the cohort overall and increased in several children before clinical relapse. Twelve-month overall survival was 40% in the cohort overall, and for children with baseline pEBV detected, survival was worse if baseline pEBV DNA was ≥6 log10 copies/mL versus <6 log10 copies/mL (p = 0.0002), and also if pEBV DNA was persistently detectable at mid-treatment versus undetectable (p = 0.041). Among children with baseline pEBV DNA detected, viremia was the only significant risk factor for death by 12 months in multivariate analyses (adjusted hazard ratio 1.35 per log10 copies/mL, 95% CI 1.04-1.75, p = 0.023). Quantitative pEBV DNA has potential utility for diagnosis, prognosis, and response assessment for pediatric BL in SSA.